Overview
A top-10 global pharmaceutical company partnered with Zelthy to deploy AI agents across its regulatory document operations. The company managed a large portfolio of regulatory documents across multiple active clinical programs in oncology and immunology, coordinating development across internal medical writing teams, CRO partners, and regional regulatory leads.
With Zelthy's AI agents and workflow platform deployed as an overlay on the client's existing Veeva environment, the company successfully transformed manual, siloed document workflows into an intelligent, automated pipeline — achieving measurable results within 12 weeks of production deployment.
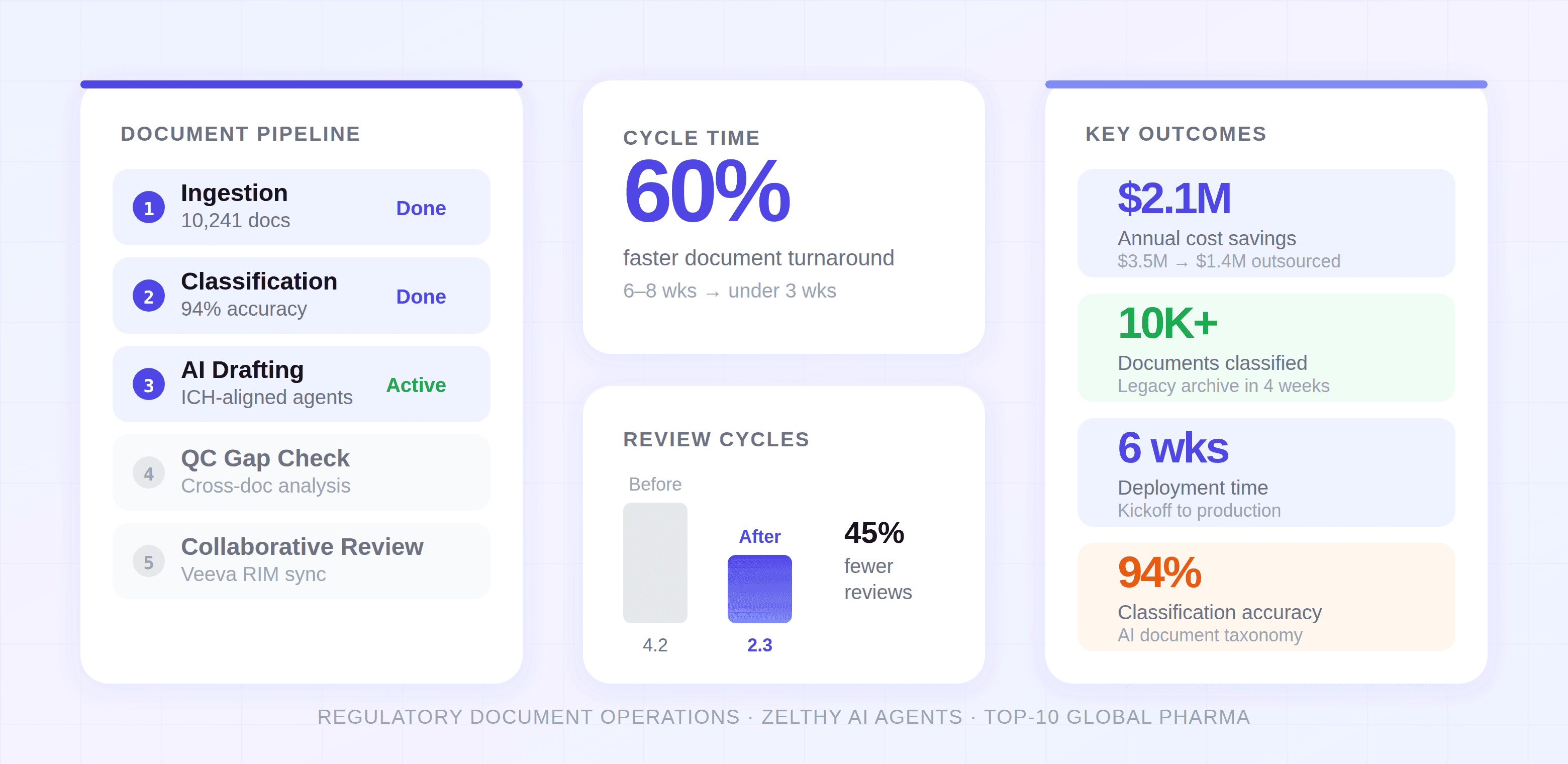
Zelthy's AI agents operate across five stages of the regulatory document lifecycle as an overlay on existing Veeva RIM environments. Document Classification agents automatically ingest, classify, and extract metadata from legacy documents, processing 10,000+ documents in under 4 weeks versus 6 months estimated for manual effort, with 94% accuracy. Drafting Acceleration agents generate structured first drafts from source documents and statistical outputs against ICH guidelines, shifting medical writers from blank-page authoring to review-and-refine workflows. Cross-Document Gap Analysis agents continuously check related documents for contradictions, missing cross-references, and terminology drift, catching issues during drafting instead of final QC. Collaborative Review workflows replace email-based tracked-changes cycles with a unified workspace maintaining full audit trails. The Veeva RIM connector syncs document status and metadata bidirectionally while maintaining 21 CFR Part 11 compliance.
Challenges
The regulatory affairs organization operated on Veeva RIM and Quality with SharePoint-based document collaboration. Annual outsourced medical writing spend exceeded $3.5M, with increasing cost pressure and no clear path to reducing that dependency. Document volume continued to grow without proportional headcount increases, and several structural problems had turned document operations into a bottleneck across the organization.
Volume and Complexity
Thousands of regulatory documents in active circulation, each with dependencies on clinical data, statistical outputs, and cross-functional inputs. Updates to source documents cascaded through dozens of downstream documents with no automated consistency checks.
Inconsistent Quality Across Vendors
Multiple CRO writing teams produced documents with varying structure, voice, and formatting standards. QC teams spent more time normalizing output than improving content, averaging 4+ review cycles per document.
Legacy Data Fragmentation
Inherited assets from multiple acquisitions existed in mixed formats — scanned PDFs, legacy Word templates, and non-standard folder structures. Classification and retrieval was almost entirely manual.
Manual Review Bottlenecks
Document review workflows relied on email threads and tracked-changes Word files circulating between authors, reviewers, and approvers. Version control was error-prone, and status visibility across the function was poor.
"Critical documents were consistently delivered late, the medical writing team was fully saturated, and the organization was spending millions on external writing support without reducing cycle times."
Solution: AI Agents Across the Document Lifecycle
Zelthy deployed a suite of AI agents and workflow tools across the regulatory document lifecycle — from ingestion and classification through drafting, review, and final quality checks. The platform was implemented as an overlay on the client's existing Veeva environment, requiring no system replacement.
AI-Powered Document Classification
Automated ingestion and classification of 10,000+ legacy documents into standardized regulatory taxonomy. AI agents extracted metadata, identified document types, mapped relationships, and flagged quality issues — replacing weeks of manual cataloging with hours of automated processing.
Drafting Acceleration Agents
AI agents configured against ICH guidelines and internal style standards generated structured first drafts from source documents, statistical outputs, and reference materials. Medical writers shifted from blank-page authoring to review-and-refine workflows, reducing time-to-first-draft significantly.
Cross-Document Gap Analysis
Continuous AI-driven consistency checks across related documents — flagging contradictions between narratives and data, identifying missing cross-references, and detecting terminology drift. Issues that previously surfaced during late-stage QC were caught at the point of authoring.
Collaborative Review Workflows
Purpose-built review environment with version control, role-based access, and structured feedback loops. Replaced email-based tracked-changes cycles with a unified workspace where medical writers, biostatisticians, and regulatory leads could collaborate in real time with full audit trails.
Veeva RIM Integration
Read-only connector syncing document status, metadata, and classification between Zelthy's workflow layer and the client's existing Veeva RIM environment. Full 21 CFR Part 11 compliance maintained throughout.
Key Outcomes & Impact
Within 12 weeks of production deployment, the regulatory operations team measured significant improvements across every stage of the document lifecycle.
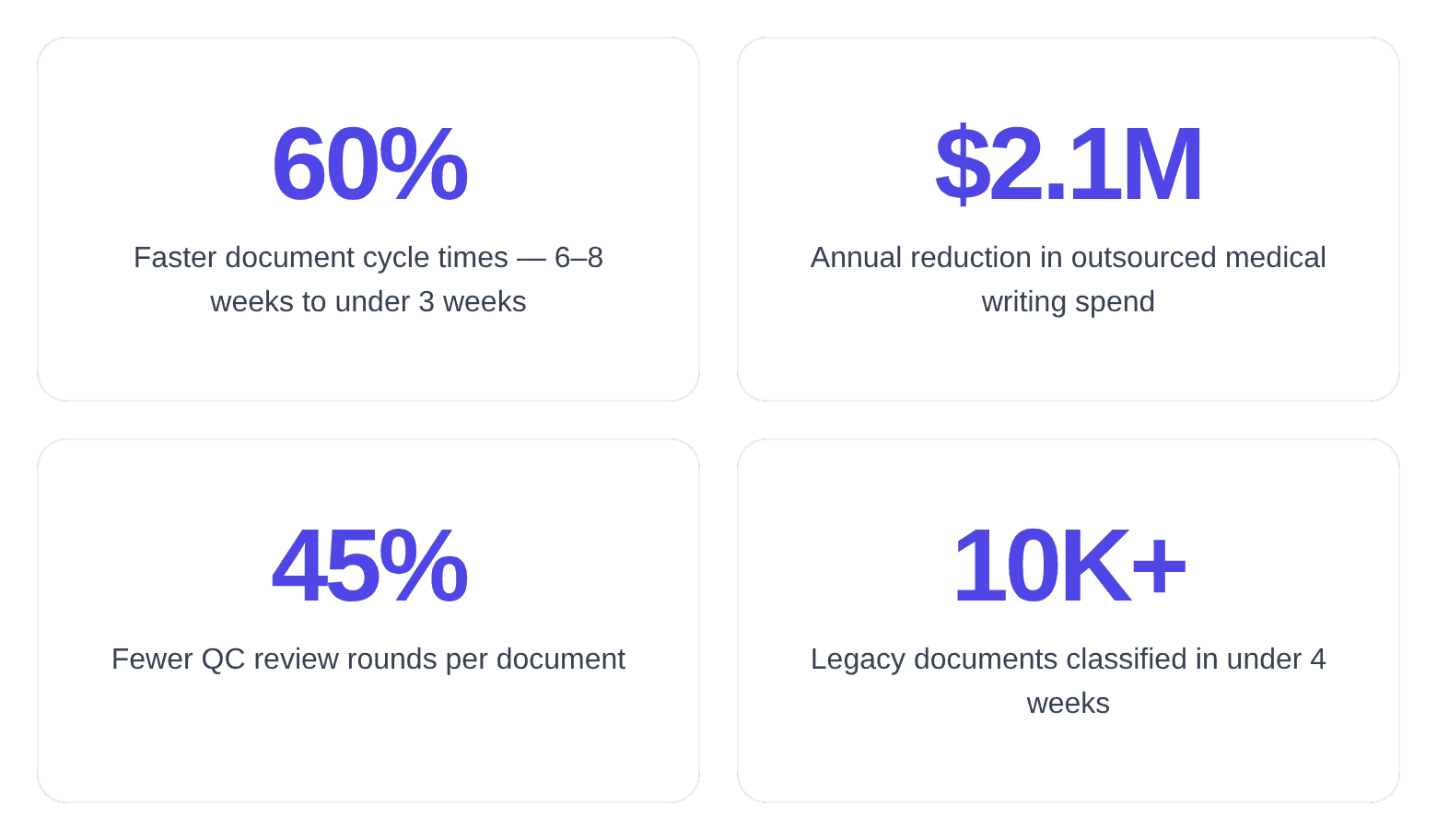
- 60% reduction in document cycle times: Average turnaround from source data availability to approved final draft dropped from 6–8 weeks to under 3 weeks. AI agents handled structural assembly and source-document integration while writers focused on scientific narrative and regulatory judgment.
- 45% fewer QC review cycles: Average review rounds per document dropped from 4.2 to 2.3. The gap analysis agent caught cross-document inconsistencies during drafting rather than during final review, eliminating the most time-consuming class of QC findings.
- $2.1M annual cost savings: Outsourced medical writing spend decreased from $3.5M to $1.4M annually. AI agents absorbed first-draft generation work previously assigned to CRO writers, while the internal team's effective capacity doubled without additional headcount.
- 10,000+ legacy documents classified in under 4 weeks: A task the team had estimated would require 6 months of manual effort. The classification engine achieved 94% accuracy on document type identification, with human review required only for edge cases.
- 6-week deployment: From kickoff to first production use. Weeks 1–2 covered legacy ingestion and Veeva connector setup. Weeks 3–4 focused on agent configuration and calibration. Week 5 ran a parallel pilot. Week 6 began full production rollout.
Conclusion
Zelthy's AI agents gave the regulatory team the capacity to handle growing document volume without growing headcount — while improving quality and reducing dependence on outsourced writing. The platform now operates as the standard document workflow layer across the client's regulatory function.
With Zelthy's configurable and scalable platform, pharmaceutical companies can rapidly deploy AI-native regulatory operations that deliver measurable improvements in speed, quality, and cost — without disrupting existing systems or workflows.
Want to learn more or see a contextual demo for your organization? Email us at connect@zelthy.com or reach us directly on LinkedIn.