Expanded access (also called compassionate use) is a regulatory pathway that allows patients with serious or life-threatening conditions to receive investigational drugs outside of clinical trials when no comparable approved therapy exists and the patient cannot enroll in an ongoing trial. In the US, the FDA administers three expanded access pathways — individual patient INDs (including emergency use approved by phone in under 24 hours), intermediate-size patient protocols, and treatment INDs for broader populations. In the EU, expanded access operates through national competent authorities under Article 83 of Regulation 726/2004, with significant variation by member state.
Expanded access programs are not clinical trials — they exist solely for treatment, not research — but they carry real regulatory obligations including IND maintenance, adverse event reporting, and IRB/ethics oversight. For sponsors, the calculus is strategic as well as ethical: FDA analysis shows that drugs with expanded access experience have a higher approval rate (84%) than those without (76%), and EA data is increasingly used in regulatory submissions and health technology assessments. This guide covers when sponsors should offer expanded access, how the US and EU frameworks differ, and the operational requirements for running a compliant program.
What is Expanded Access?
Expanded Access (also known as Compassionate Use) is a U.S. FDA and EU regulatory framework that enables patients to obtain investigational drugs when:
- No comparable or satisfactory therapy is available.
- The patient cannot join an ongoing clinical trial.
- The potential benefit outweighs potential risks.
Key Difference from Clinical Trials: Clinical trials generate safety and efficacy data under controlled conditions, while expanded access is strictly for treatment purposes, not research.
Right-to-Try legislation (2018) provides a separate pathway, allowing access without FDA oversight or IRB review, but requires that the drug has completed Phase I trials and that the manufacturer agrees.
When Should Sponsors Offer Expanded Access?
1. Patient Eligibility Criteria
Sponsors may offer EA when:
- Patients have serious or immediately life-threatening diseases.
- No standard or alternative treatment options exist.
- The patient cannot participate in available clinical trials.
- The potential benefit justifies potential risks.
Many sponsors manage expanded access alongside broader patient support operations. Zelthy's patient services platform handles both, with enrollment, eligibility screening, adherence monitoring, and engagement workflows on a single platform.
2. Development Stage Requirements
EA pathways accommodate various development stages, though specific requirements vary by program type.
- Individual Patient Access: Generally requires Phase I safety data.
- Intermediate-size populations: Need stronger safety evidence.
- Treatment INDs (widespread use): Require substantial safety and efficacy data.
3. Safety Data Prerequisites
Sponsors must provide safety data showing:
- Known dose-limiting toxicities
- Possible drug–drug interactions
- Contraindications
The FDA evaluates whether “risks are not unreasonable given the disease severity.”
4. Regulatory Triggers
- United States: FDA authorization is required except Right-to-Try pathways. Emergency requests can be approved by phone in <24 hours.
- European Union: National competent authorities decide, with EMA offering coordination for multi-country programs.
How Should Sponsors Offer Expanded Access?
U.S. FDA Process
Three main EA pathways exist under FDA:
- Individual patient INDs (including emergency use).
- Intermediate-size patient protocols.
- Treatment INDs for broad populations.
- Emergency use: FDA approval by phone; IRB notified within 5 days.
- Non-emergency: IND + IRB approval required; typical review time ~4 days.
EU Process
European programs operate through national competent authorities under Article 83 of Regulation 726/2004. The EMA provides harmonized recommendations, but implementation varies by member state.
For example, countries like:
- France: Temporary Authorization for Use (ATU).
- UK: Early Access to Medicines Scheme (EAMS) with real-world data collection.
Documentation and Reporting
- US sponsors must maintain current INDs with protocol amendments for any changes, annual reports, and expedited safety reporting within 15 days of serious, unexpected, related adverse events.
- EU reporting requirements vary significantly by country, with some requiring periodic efficacy and safety updates.
These safety monitoring and audit trail requirements are part of a broader compliance landscape. Zelthy's compliance and quality platform automates adverse event lifecycle management, inspection readiness, and continuous GxP documentation.
Patient Consent and Ethics
- US: IRB approval is mandatory for all US EA programs except emergency use, where post-treatment notification within five days suffices.
- EU: Some countries (e.g., Italy, Spain) require ethics review; others delegate responsibility to physicians.
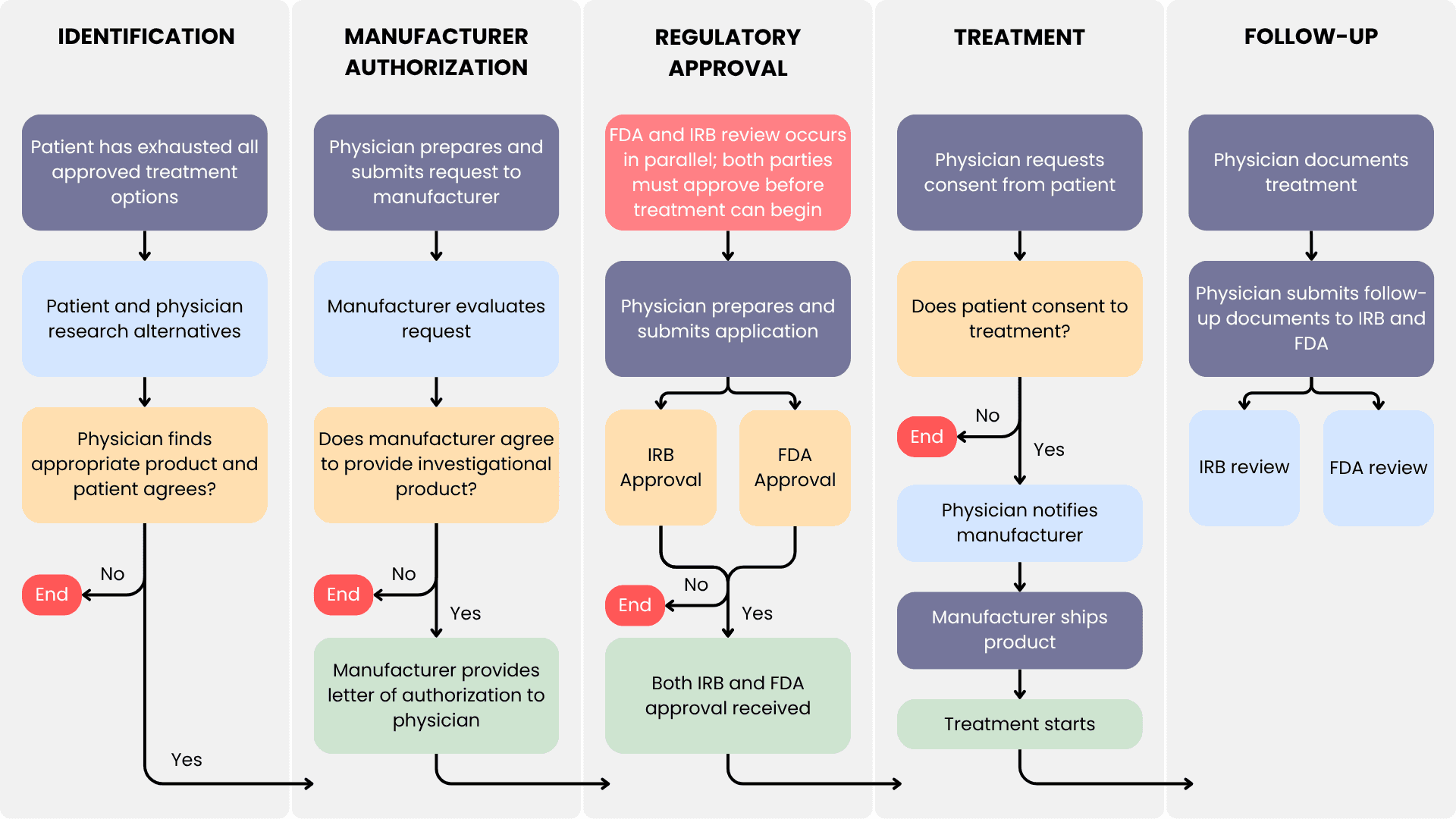
Expanded Access Request Process (Single Patient)
The single-patient expanded access process begins when a treating physician identifies a patient with a serious or life-threatening condition who cannot participate in available clinical trials. For emergency use, the FDA can authorize access by phone in under 24 hours, with IRB notified within 5 days. For non-emergency requests, both IND submission and IRB approval are required, with typical FDA review taking approximately 4 days. The sponsor must provide current safety data and maintain the IND with protocol amendments, annual reports, and expedited safety reporting for serious adverse events within 15 days.
U.S. vs EU: Expanded Access vs Compassionate Use

US expanded access operates under FDA oversight through three pathways: individual patient INDs (including emergency use with phone authorization in under 24 hours), intermediate-size patient protocols, and treatment INDs for broad populations. All require IND submission and IRB approval (except emergency use, where IRB is notified post-treatment within 5 days).
EU compassionate use operates through national competent authorities under Article 83 of Regulation 726/2004, with implementation varying by member state — France uses Temporary Authorization for Use (ATU), the UK offers the Early Access to Medicines Scheme (EAMS) with real-world data collection, and countries like Italy and Spain require ethics review while others delegate to physicians.
Both regions require safety monitoring and adverse event reporting, but US reporting obligations are more standardized (15-day expedited reporting for serious events) while EU requirements vary significantly by country.
These US–EU regulatory differences mirror the broader divergence in how each region designs patient support. Our US vs EU PSP comparison maps these structural differences across the full patient services landscape.
Benefits of Expanded Access for Sponsors
- Patient goodwill & reputation: EA programs demonstrate commitment to patient access and can enhance relationships with physician communities and patient advocacy groups.
- Real-world evidence: EA data increasingly used in regulatory submissions and health technology assessments (HTA).
- Regulatory advantage: FDA analysis shows drugs with EA have higher approval rates (84%) than those without (76%).
Risks and Considerations
- Resource demands: Represents the primary operational challenges. Regulatory, medical, and supply chain teams must manage EA alongside trials.
- Manufacturing capacity: Small biotechs may struggle to supply both EA and trials.
- Regulatory obligations: These include ongoing safety monitoring, adverse event reporting, and protocol maintenance. However, FDA analysis of 321 regulatory decisions found no instances where EA experience led to negative approval decisions.
Here’s How Zelthy Supports Expanded Access Programs
Managing Expanded Access requires precise coordination across regulatory submissions, patient eligibility tracking, safety reporting, and supply management. Many sponsors—especially emerging biopharma—struggle to balance these demands alongside pivotal clinical trials.
Zelthy’s digital health platform simplifies this process by:
- Streamlining regulatory workflows: Centralized tracking of IND submissions, IRB approvals, and country-specific documentation.
- Enabling real-time patient and physician engagement: Secure portals for request intake, eligibility screening, and consent management.
- Automating compliance reporting: Built-in tools for safety event capture, monitoring, and timely regulatory submissions.
- Scaling globally: Configurable modules that adapt to the U.S. FDA Expanded Access, EU Compassionate Use, and other country-specific frameworks.
By integrating operational, regulatory, and patient-facing functions into a single platform, Zelthy enables sponsors to expand patient access without straining resources—while maintaining compliance, transparency, and data integrity.
Expanded access management is one module within Zelthy's broader regulatory operations platform — which also covers submission pipeline management, dossier assembly, regulatory intelligence, and labelling management across jurisdictions.
A leading Australian pharmaceutical company partnered with Zelthy to digitize its Compassionate Access Program (CAP), cutting approval times by 65%, reducing costs by 40%, achieving 99% compliance with automated audit trails, improving real-time visibility for stakeholders, and strengthening supply chain forecasting—ultimately delivering critical therapies faster and more efficiently.
Read the full story to find out how they managed to cut approval times from 7–10 days to 2–3 days by digitizing their compassionate access program while maintaining full regulatory compliance.
Book a demo and talk to us about building your Expanded Access framework with Zelthy. Contact us at connect@zelthy.com or send us a DM on LinkedIn.
Frequently Asked Questions
What is expanded access (compassionate use)?
Expanded access, also called compassionate use, is a U.S. FDA and EU regulatory pathway allowing patients with serious or life-threatening diseases to access investigational drugs outside of clinical trials when no approved treatment exists. Unlike clinical trials, which generate safety and efficacy data under controlled conditions, expanded access is strictly a treatment pathway for patients who cannot enroll in a trial and have no other options.
What is the difference between expanded access and Right-to-Try?
Expanded access requires FDA authorization and, for non-emergency cases, IRB approval before a patient can receive an investigational drug. Right-to-Try legislation (2018) allows access without FDA oversight or IRB review, provided the drug has completed Phase I trials and the manufacturer agrees to supply it. Right-to-Try removes regulatory gatekeeping but also removes the FDA's patient safety oversight and adverse event monitoring requirements that apply to expanded access programs.
When should a sponsor offer expanded access?
Sponsors may offer expanded access when a patient has a serious or immediately life-threatening disease, no comparable approved treatment exists, the patient cannot enroll in an available clinical trial, and the potential benefit justifies the risks. Individual patient access generally requires Phase I safety data. Intermediate-size and Treatment IND programs require progressively more robust safety and efficacy evidence before the FDA will authorize broad access.
How fast can the FDA approve an emergency expanded access request?
For emergency individual patient requests, the FDA can grant approval by phone in under 24 hours. The sponsor submits the IND and IRB notification follows within five days of treatment. For non-emergency individual patient requests, typical FDA review time is approximately four days. This speed reflects the agency's recognition that patients with immediately life-threatening conditions cannot wait for standard regulatory timelines.
Does running an expanded access program affect a drug's approval chances?
FDA analysis of 321 regulatory decisions found no instances where expanded access experience led to a negative approval outcome. In fact, drugs with expanded access programs have a higher approval rate (84%) than those without (76%), likely because real-world EA data supplements the clinical evidence package for health technology assessments and regulatory submissions. The primary concern for sponsors is operational, resource demands on regulatory, medical, and supply chain teams running EA alongside pivotal trials.
How does expanded access data contribute to regulatory submissions?
Since 2017, the FDA has approved multiple drugs based on real-world evidence, and expanded access data increasingly contributes to this evidence base. EA data, capturing treatment outcomes, adverse events, and dosing patterns outside trial conditions, supports HTA submissions, post-market safety reporting, and comparative effectiveness arguments in payer negotiations. Sponsors who implement structured data collection in EA programs, rather than treating them as purely operational, generate regulatory and commercial value from patient access commitments.
How do EU compassionate use programs differ from the US expanded access pathway?
In the EU, compassionate use is governed by national competent authorities under Article 83 of Regulation 726/2004, meaning program structure and approval timelines vary by country. France uses Temporary Authorizations for Use (ATU); the UK operates the Early Access to Medicines Scheme (EAMS). The EMA provides harmonized recommendations but does not centrally authorize programs. By contrast, the US FDA runs a unified expanded access framework with standardized IND submissions and defined review timelines applicable across all states.
References
- Office of the Commissioner. (2025, September 8). Expanded access. U.S. Food And Drug Administration.
- Jarow, J. P., Lurie, P., Ikenberry, S. C., & Lemery, S. (2017). Overview of FDA’s expanded access program for investigational drugs. Therapeutic Innovation & Regulatory Science, 51(2), 177–179. doi:10.1177/2168479017694850
- Ferdman, J. M., Peloquin, D., Ropes & Gray LLP, Bierer, B. E., & Multi-Regional Clinical Trials Center of Brigham & Women’s Hospital and Harvard. (2025). New developments in right to try legislation. [pdf]
- Klopfenstein, M., Van Campen, L. E., & Garnett, T. (2015). Expanded Access Programs: ethical and practical considerations for biopharmaceutical sponsors. Therapeutic Innovation & Regulatory Science, 49(3), 352–358. doi:10.1177/2168479015578154
- U.S. Food And Drug Administration (2017). Expanded Access to Investigational Drugs for Treatment Use — Questions and Answers Guidance for Industry. [pdf]
- European Medicines Agency (EMA) (2020). Compassionate use | European Medicines Agency (EMA).
- Whitfield, K., et al. (2010). Compassionate use of interventions: results of a European Clinical Research Infrastructures Network (ECRIN) survey of ten European countries. Trials, 11(1). doi:10.1186/1745-6215-11-104
- Holbein, M. E. B. (2009, August 1). Understanding FDA regulatory Requirements for investigational new Drug Applications for Sponsor-Investigators. [pmc]
- Polak, T. B., Van Rosmalen, J., & De Groot, C. a. U. –. (2020). Expanded Access as a source of real‐world data: An overview of FDA and EMA approvals. British Journal of Clinical Pharmacology, 86(9), 1819–1826. doi:10.1111/bcp.14284
- Wasser, J. S., & Greenblatt, D. J. (2023). Applying real-world data from expanded-access (“compassionate use”) patients to drug development. Journal of Clinical and Translational Science, 7(1). doi:10.1017/cts.2023.606
- Fountzilas, E., Said, R., & Tsimberidou, A. M. (2018). Expanded access to investigational drugs: balancing patient safety with potential therapeutic benefits. Expert Opinion on Investigational Drugs, 27(2), 155–162. doi:10.1080/13543784.2018.1430137
- Jarow, J. P., & Moscicki, R. (2017). Impact of expanded access on FDA regulatory action and product labeling. Therapeutic Innovation & Regulatory Science, 51(6), 787–789. doi:10.1177/2168479017707800
- Jarow, J. P., et al. (2016). Expanded Access of Investigational Drugs: The experience of the Center of Drug Evaluation and Research over a 10-Year period. Therapeutic Innovation & Regulatory Science, 50(6), 705–709. doi:10.1177/2168479016656030


![AI Output Compliance Monitoring in Pharma: The Watch Layer [2026]](/_next/image?url=https%3A%2F%2Fhumble-friendship-ab99f71d93.media.strapiapp.com%2FAI_Compliance_in_Pharma_a76c884d28.png&w=3840&q=75)
