The pharmaceutical compliance stack was built for a world that no longer exists. Quarterly sampling of HCP interactions. Manual aggregation of spend data across Concur, Cvent, and grants systems. Narrative risk assessments filed at committee. SOP updates that lag regulatory change by weeks. That stack was designed for a regulatory environment that was inspected episodically. It is now being enforced against continuously.
OPDP is surveilling digital promotional content at a pace no sampling regime can match. EFPIA's Code of Practice amendments are tightening year over year. FDA has signalled expectations for AI output monitoring. India's DPDP Act reshapes the data basis on which most PSPs were designed. The enforcement surface is 100%. The monitoring surface, in most pharma companies, is still 2–5%, the transactions you sampled.
This is the gap. Closing it does not mean buying a bigger rules engine or writing stricter SOPs. It means building compliance on a different substrate; one that reads everything, understands context, and surfaces what matters. That is what an AI-powered compliance platform does.
Zelthy is an AI-powered compliance platform for life sciences operations, built on open standards for regulated industries. It serves 6 of the top 10 global pharma companies; including Roche, Bristol-Myers Squibb, AstraZeneca, MSD, Novartis, and Servier, with compliance deployments across 12+ countries and 45+ live compliance applications in production.
This piece is for compliance officers, commercial operations heads, and digital leaders deciding what the next compliance stack should look like. It argues the category, AI-powered compliance platforms for life sciences, is real and necessary, walks through the six capabilities a credible platform must have, frames a buyer's evaluation checklist, and is honest about where platforms like Zelthy fit, and where they don't.
The compliance stack that's breaking down
Walk through how a typical top-10 pharma monitors HCP engagement compliance today. Field teams log interactions in Veeva CRM. Expenses flow through SAP Concur. Speaker programs and congress attendance sit in Cvent. Grants and medical education move through a separate grants platform, sometimes Steeprock, sometimes a homegrown system. Promotional materials route through a PromoMats workflow. Each of these systems is well-governed individually. The compliance team reconciles them quarterly, usually by pulling exports, normalizing them in Excel or a reporting layer, and sampling 2–5% of transactions for review against SOPs.
Regulatory intelligence runs on a parallel track. An OIG advisory drops. An EFPIA Code amendment is published. FDA releases guidance on AI-generated promotional content. A compliance analyst reads it, maps it mentally to the policies and programs that might be affected, opens a change request, and routes it through policy review. From publication to updated SOP to retrained staff, six to twelve weeks is typical.
None of this is incompetence. It is what the stack was designed to do when enforcement was episodic. The problem is that enforcement is no longer episodic. OPDP monitors digital promotional content in near-real-time. The Sunshine Act covers 100% of transfers of value. India's DPDP Act requires demonstrable consent lineage on every patient touchpoint, not an audit-time narrative. The HHS Office of Inspector General has progressively raised expectations for self-monitoring across the pharma sector.
The math breaks. If 2% of transactions are reviewed and 100% are enforced against, the remaining 98% is an unmonitored surface. Inspection findings increasingly come from that 98%, the interactions compliance never looked at, the grants decisions that didn't cross the review threshold, the speaker programs whose attendee lists drifted from the original approval.

This is the gap a compliance intelligence platform is built to close. Not by adding one more reporting dashboard on top of the existing stack, but by continuously reading the raw transaction data across every source, applying policy-aware AI agents to classify what it sees, and escalating only what is genuinely ambiguous to a human reviewer. The model flips from sampling to continuous assurance.
Why rules engines can't scale
The deeper argument is this: for the last twenty years, pharma has treated compliance as a process problem. Tighter approval workflows, more thorough risk committees, better SOP libraries. The response to every enforcement action has been to add a rule, add an approval step, add a reviewer. The result is the stack described above, well-governed in the small, blind in the aggregate.
Compliance is not fundamentally a process problem. It is an intelligence problem. The question a monitoring system has to answer is: of the hundreds of thousands of transactions moving through our commercial systems this quarter, which ones are evidence of a compliance risk? Rules engines answer this by encoding someone's prior understanding of what risk looks like. If the rule exists, the transaction gets flagged. If the rule doesn't exist, because the interaction pattern is new, the program type is new, the geography is new, the transaction is invisible.
Every enforcement action in the last five years has revealed a gap the rules library didn't cover. Speaker programs that looked compliant by policy but violated the underlying statute. Grant decisions within thresholds but clustered in ways that telegraphed intent. PSP data flows that satisfied HIPAA but failed DPDP. Digital promotional claims approved through MLR but drifted in social syndication. None would have tripped a rule, because the rule was written before the pattern existed.
AI agents approach the problem inversely. Rather than encoding what a violation looks like and waiting for a match, they read the raw signal, the transaction, the interaction log, the policy corpus, the regulatory text, and classify each instance in context. A prescription for an off-label indication is not flagged because it matches a rule; it is flagged because the model understands, from the clinician's history, the product label, and the policy corpus, that the pattern is ambiguous enough to warrant review. The output is not a list of alerts. It is a ranked queue of signals, each with context and a recommended compliance lane.
IQVIA SmartSolve and IONI.ai are exploring this same insight from different angles - building AI layers on top of existing compliance workflows. Veeva Vault's AI add-ons are doing the same for document-centric processes. The distinction Zelthy makes is architectural: we did not retrofit AI onto a workflow engine. The agents, the policy corpus, and the operational systems were built into the platform from day one, on an open-source framework designed for regulated industries.
Six capabilities of an AI-powered compliance platform
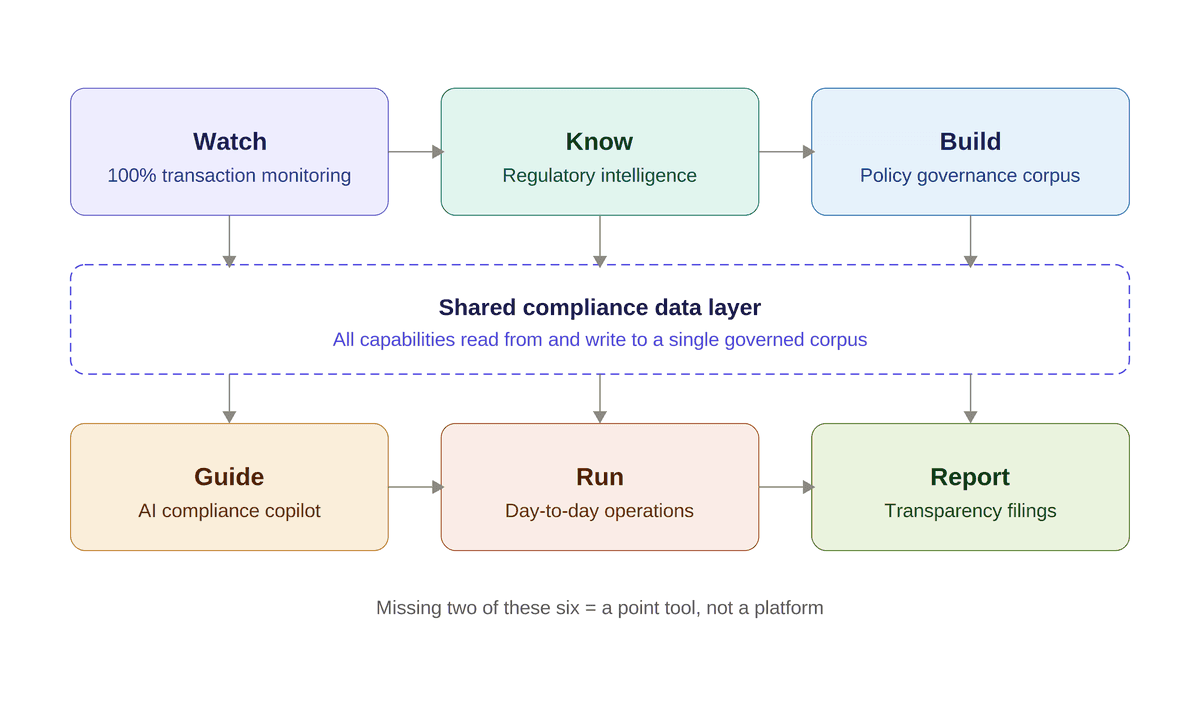
A compliance intelligence platform is defined by six capabilities. They are distinct; each has its own data model and its own AI architecture. But they compose — one layer feeds the next. Any platform missing two of these is a point tool dressed up in category language.
Watch — continuous monitoring at the transaction level
Watch is the monitoring layer. It reads every HCP interaction, every transfer of value, every grant decision, every program execution event, not a sample. AI agents classify each transaction against the policy corpus and the regulatory baseline, scoring it for compliance risk. Ambiguous signals route to the right compliance lane; commercial compliance, medical compliance, privacy, transparency, with context and the relevant SOP citation.
At a top-5 global oncology company, Zelthy's continuous monitoring agents moved compliance from quarterly HCP interaction sampling to 100% real-time scanning across eight markets, flagging deviations with context and routing them to the right compliance lane within hours instead of quarters. The audit package stays current year-round, not compiled in a three-week sprint before inspection. The coverage change is not incremental. Going from 2% to 100% changes what compliance as a function can credibly say about the state of the business.
See also: AI output compliance monitoring in pharma, a deeper treatment of how AI-generated content itself is monitored for regulatory alignment, which operates as a specialised layer within Watch.
Know — regulatory intelligence as impact mapping
Know converts regulatory change into operational work. A new OIG advisory publishes. An EFPIA amendment clears. FDA releases a new guidance on promotional review. The Know layer ingests the text, identifies the policy dimensions it affects, and maps it to the specific SOPs, programs, training modules, and attestations in the client's corpus. Output is not a summary email. It is a list of affected artifacts, owners, and recommended actions, typically within minutes of publication. Impact analysis that used to take a week is 90% faster.
Build — policy governance as a living corpus
Build is the authoring and governance layer. Policies, SOPs, training decks, attestation campaigns, and risk configurations live under a single lifecycle. When a policy changes, the downstream artifacts, training modules, attestation cycles, the knowledge base the Guide copilot draws from, propagate the change automatically. When a policy retires, the dependencies retire with it. The corpus is versioned, traceable, and auditable at any point in time. This is what makes the AI copilot credible: the policy corpus the AI cites is the policy corpus the business is operating under, not a snapshot from six months ago.
Guide — AI copilot grounded in your policy corpus
Guide is the AI compliance copilot — the interface a field rep, a medical liaison, or a compliance analyst uses to get an answer in 30 seconds instead of a 30-minute email thread. Unlike a generic GRC chatbot, it cites the specific SOPs in force for that person's role, market, and program. It does not generate plausible-sounding compliance answers; it grounds every response in the Build corpus and surfaces the citation. When a question is genuinely ambiguous; a fair market value question on an unusual speaker fee, a nuanced grant eligibility scenario, it routes the question to the correct compliance lane with the context already assembled.
Run — purpose-built operational systems
Run is the day-to-day operational software the compliance team uses: audit management, CAPA tracking, conflict of interest disclosures, outside activity approvals, compliance calendar, training attestations. These are configured, not custom-built. A global pharma deploys them in 4–8 weeks on the Zelthy platform, typically replacing four to six disparate internal applications. Each Run system writes to the same compliance corpus the Watch agents monitor and the Guide copilot cites, which is what makes the whole stack coherent rather than federated.
Report — regulatory filings across every jurisdiction
Report is the transparency reporting layer. US Sunshine Act Open Payments. EFPIA Code Disclosure across 33 European markets. France Loi Bertrand. UK ABPI. Japan JPMA. Australia Medicines Australia. Each jurisdiction has its own taxonomy, threshold, and submission format. The Report layer aggregates transfers of value continuously from the source systems, normalizes to each jurisdiction's schema, and maintains submission-ready packages year-round, not assembled in a quarter-end sprint. Filing becomes a review-and-submit act, not a reconstruction.
A buyer's evaluation framework
Five questions to ask every vendor in this category, not just Zelthy. These are the questions that separate a compliance intelligence platform from a workflow engine with "AI" in the marketing.
1. Does the AI cite your actual policies, or generate plausible-sounding answers? What to look for: every AI response shows the citation, SOP number, version, section, and the cited document is the version currently in force. Red flag: the vendor demos an AI copilot with generic pharma answers rather than answers grounded in a client-specific policy corpus. If the model can't show where the answer came from, it is hallucinating.
2. Does monitoring cover 100% of transactions, or sample a subset? What to look for: the platform reads every row in the source system, not a configured sample. Ask for the architecture, stream processing, batch ingestion, query-on-demand, and the throughput in transactions per day at the largest deployment. Red flag: coverage described in terms of "dashboards" or "reports" rather than transaction volume actually being classified.
3. When a regulation changes, how does the platform identify impacted SOPs, programs, and owners? What to look for: a demonstration, not a slide. The vendor should be able to show a recent regulatory change and walk through the output, the list of SOPs flagged, the programs affected, the attestation cycles triggered, the owners notified, with timestamps. Red flag: the answer is "we send a regulatory intelligence digest." A digest is not impact analysis.
4. Can your internal engineering read and modify the platform code, or is it a vendor black box? What to look for: platform source code the client can audit, extend, and fork. For Zelthy, this is the Zango framework, open source under a standard license. Red flag: configuration layers on top of a closed platform where every customization is a billable vendor engagement. Vendor lock-in in compliance technology is a structural risk, not a commercial one; it limits how fast you can respond to the next regulatory shift.
5. How does the platform integrate with Veeva, SAP Concur, Cvent, and the grants systems already in production? What to look for: named connectors with a maintenance history. Ask which integration broke most recently and how long resolution took. Red flag: "we support any system via API." Generic API support means every integration is a custom build and every vendor upgrade is a surprise.
Where AI-powered compliance platforms fit — and where they don't
This is the posture. A compliance intelligence platform is not a replacement for every system in the regulated tech stack. Understanding the boundary makes the claim within the boundary credible.
If you need document management for regulated documents, controlled, versioned, eSignature-ready, that is Veeva Vault. If you need pharmacovigilance case intake and processing, that is Oracle Argus or Veeva Safety. If you need enterprise GRC across finance, HR, IT, and legal, the full SOX-adjacent scope, that is NAVEX, LockPath, MetricStream, or SAI360. If you need a Quality Management System for GMP manufacturing, that is MasterControl or ComplianceQuest. These are mature categories with incumbent leaders, and nothing in the AI-powered compliance platform category should be understood as a replacement for them.
Zelthy is the compliance intelligence and operations layer you build on top, specifically for commercial and medical-affairs compliance workflows the enterprise tools above don't cover natively. Continuous HCP interaction monitoring. Program governance at the brief stage. Transfer-of-value aggregation for transparency reporting. The AI compliance copilot grounded in the field-facing SOP corpus. The operational systems, audit, CAPA, COI, that the compliance team runs day to day.
The comparator most pharma companies evaluate against is Veeva Compliance Suite, the closest thing in the incumbent stack to what's described here. The difference is implementation time and architecture: Zelthy is modular, AI-native, and deployed in 4–8 weeks per capability, on open-source infrastructure the client can own. Veeva Compliance Suite is a 12–18 month enterprise implementation on closed infrastructure. Both are defensible depending on scope and risk profile. Being honest about which problem each solves better is how this category becomes credible.
Implementation pathway
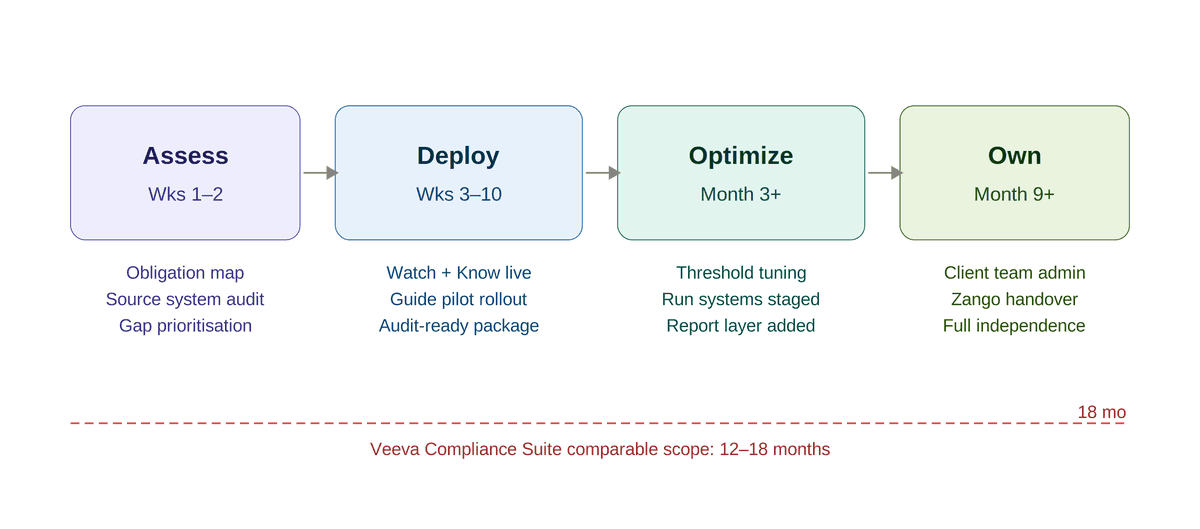
How a pharma compliance function gets from the current state to an operational AI-powered compliance platform in 2026. Four phases.
Phase 1: Assess (1–2 weeks). Inventory current obligations: jurisdictions, transparency reporting thresholds, inspection history, known SOP gaps, active consent decrees or CIAs. Map the source systems; Veeva CRM, Concur, Cvent, grants, PromoMats, and the current reconciliation flows. Identify the two or three capabilities that close the largest audit gap first. For most pharma companies this is Watch and Know.
Phase 2: Deploy (4–8 weeks). Configure the selected capabilities against the actual policy corpus. Ingest historical transactions for model baselining. Connect the source systems through standard connectors. Deploy the Guide copilot for a pilot population, typically commercial compliance analysts and a subset of field teams, to calibrate response quality before broader rollout. Run Watch in parallel with the existing sampling process for two cycles, then cut over.
Phase 3: Optimize (ongoing). Tune classification thresholds to reduce false positives. Expand coverage to adjacent programs. Layer in Run systems, audit, CAPA, COI, one at a time, each inheriting the policy corpus already in Build. Begin Report layer for upcoming transparency filing cycles.
Phase 4: Own (progressive). Transition platform administration to the client's compliance engineering team or a captive GCC. The open-source Zango foundation means this transition is an operational transfer, not a vendor-permission event. Most clients reach independent administration between months nine and eighteen.
At a global top-10 pharma, this sequence ran six weeks from kickoff to the first audit-ready transparency package across 12 markets. Veeva Compliance Suite on the same scope is typically a 12–18 month implementation. Custom builds on enterprise GRC platforms run longer still and start depreciating before they go live, because the regulatory environment has already moved.
What 2027 compliance looks like
Four directions of travel, not predictions, just what the market is moving toward.
Agentic compliance moves from monitoring to acting, within constraints. Today's AI agents read, classify, and escalate. The next step is constrained action; reverting a non-compliant promotional asset, rejecting a grant request that fails eligibility, pausing an approval workflow until the required attestation is captured. The human compliance lane stays in the loop; the agent does the work the lane would have done manually.
Policy corpora becomes queryable by field teams in natural language. "Am I allowed to do this" is the question every commercial person has and rarely asks because the channels are slow. A copilot grounded in the actual corpus collapses that question from a 30-minute email thread to a 30-second answer.
Regulatory intelligence arrives as actionable work, not reading material. The analyst reviews rather than authors. This is the 90% efficiency gain, already in production at the deployments above.
The compliance function shifts from reactive audit-prep to proactive continuous assurance. Inspection readiness stops being a quarterly project. The audit package is current because the monitoring is current. The function can finally spend time on the programs that are about to be launched rather than the programs that are already ran.
Frequently Asked Questions
What is an AI-powered compliance platform for life sciences?
An AI-powered compliance platform for life sciences continuously monitors HCP interactions, grants, transparency data, and program execution events using AI agents, maps regulatory change to a managed policy corpus, and operates the day-to-day compliance systems on a single governed data layer. Zelthy is one example, deployed across 6 of the top 10 global pharma companies in 12+ countries.
How does AI help pharma companies manage regulatory change?
AI ingests new regulatory publications, OIG advisories, EFPIA Code amendments, FDA guidance, and maps each to the specific SOPs, programs, and owners it affects in a client's policy corpus. Impact analysis that used to take a compliance analyst a week now runs in minutes, with an explicit owner-and-action list rather than a summary digest.
What is continuous compliance monitoring in pharma?
Continuous compliance monitoring reads 100% of relevant transactions, HCP interactions, transfers of value, grant decisions, against policy in real time, rather than sampling 2–5% quarterly. The outcome is that the audit package stays current year-round and inspection findings stop coming from the 95% of activity that was previously unmonitored.
What is an AI compliance copilot and how does it work?
An AI compliance copilot is a natural-language interface grounded in a client's actual policy corpus. It retrieves the specific SOP passages relevant to the role, market, and program, and returns a cited answer in under 30 seconds. Every response is traceable back to the source policy currently in force.
How long does it take to deploy a pharma compliance platform?
On an AI-native platform, a first capability (Watch or Know) typically goes live in 4–8 weeks. A comparable deployment on Veeva Compliance Suite runs 12–18 months. Full six-layer coverage is usually staged over 6–9 months, one capability at a time.
How do pharma companies manage Sunshine Act and EFPIA transparency reporting?
Leading compliance teams aggregate transfers of value continuously from Concur, Cvent, grants platforms, and CRM systems, normalize to each jurisdiction's schema, US Sunshine Act, EFPIA Code Disclosure across 33 European markets, Loi Bertrand, ABPI, JPMA, Medicines Australia, and maintain submission-ready packages year-round. Continuous aggregation is how filing becomes a review act rather than a quarter-end reconstruction.
See it running
Book a call with the Zelthy compliance team. We'll show you a monitoring agent running inside a live multi-market PSP, walk through the policy corpus and Guide copilot for a comparable therapeutic area, and map your current compliance landscape to a staged 4–8 week pilot scope. Typical turnaround from first call to pilot plan: two weeks.

![AI Output Compliance Monitoring in Pharma: The Watch Layer [2026]](/_next/image?url=https%3A%2F%2Fhumble-friendship-ab99f71d93.media.strapiapp.com%2FAI_Compliance_in_Pharma_a76c884d28.png&w=3840&q=75)

